Hi All,
After viewing many ideas for rust removal, I have decided to have a go at electrolysis. I have been very impressed with the articles and results that have been posted on this subject.
For my build I have tried to do this as cheaply as possible. The bucket and power supply were purchased from eBay, bucket priced at £1 and the power supply £12. The control box was from Maplin priced at £2 and the soda crystals £1 from supermarket. The wire crimps are a few pence each. The Anodes are cut from a piece of black bar found in a skip and all other parts were salvaged items from scraped finds.
The power supply is 240V AC input 12V DC output.
Back of control box
Front of control box
Photo showing bucket with six anodes linked together
Six steel anodes (+) and cathode (-) across bucket.
Complete set up. NOTE: The lid will not be in place when the process is taken place, when not in use all items can be stored in side the bucket and the lid will keep eveything together.
Thank you for viewing. I will post the results of first rust removal/clean up when completed.
The Home Engineer.


 LinkBack URL
LinkBack URL About LinkBacks
About LinkBacks








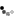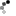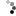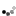
 Reply With Quote
Reply With Quote

Bookmarks